Ocugen’s Vaccine Scientific Advisory Board said,
“The COVID-19 pandemic has caused unmatched devastation to individual patients and to the world. It is going to take the kind of unmatched collaboration and innovation that is occurring right now to effectively fight back. Vaccines such as COVAXIN that can potentially elicit a broad immune response and may limit future COVID-19 severity could be important to have in our arsenal.”
“The COVID-19 pandemic has caused unmatched devastation to individual patients and to the world. It is going to take the kind of unmatched collaboration and innovation that is occurring right now to effectively fight back. Vaccines such as COVAXIN that can potentially elicit a broad immune response and may limit future COVID-19 severity could be important to have in our arsenal.”
|
Because of the COVID-19 Pandemic:
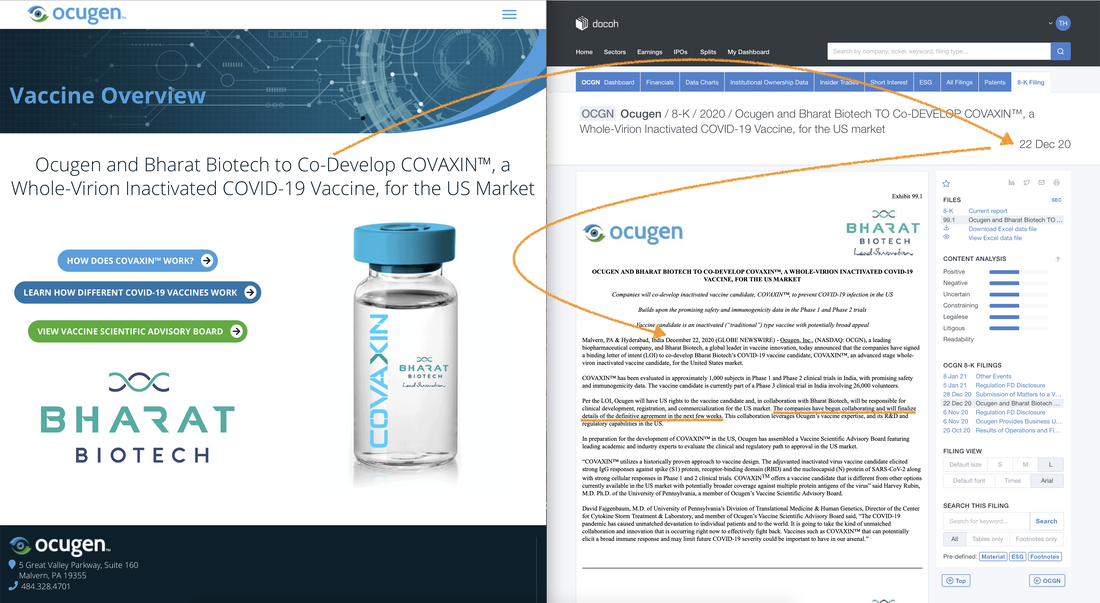
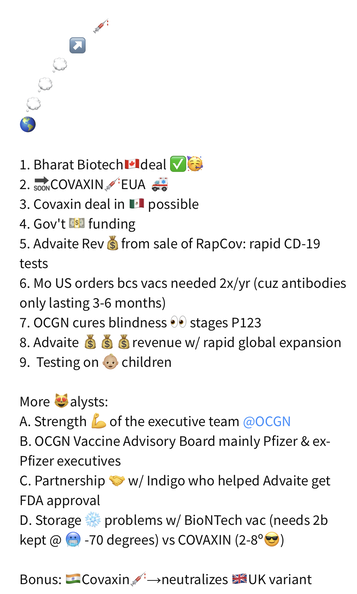
Ocugen and Bharat Biotech are to Co-Develop COVAXIN (a Whole🦠Virus Inactivated COVID-19 Vaccine) for the US Market. Building upon the promising safety and immunogenicity data in the Phase 1 & Phase 2 trials, this vaccine candidate is an inactivated "traditional" type vaccine with potentially broad appeal. |
Corporate Profile
Ocugen, Inc. is a biopharmaceutical company focused on discovering, developing and commercializing transformative therapies to treat rare and underserved eye diseases. Our breakthrough modifier gene therapy platform has the potential to treat multiple retinal diseases with one drug – “one to many” and our novel biologic product candidate aims to offer better therapy to patients with underserved diseases such as wet age-related macular degeneration, diabetic macular edema and diabetic retinopathy. |
The next catalyst coming early to mid-March
A Summary of the next 3 Big Steps:
1. Going from the binding Letter of Intent (LOI) to the Definitive Agreement (DA) which should materialize at anytime. ✅ DONE 😉
2. Completing and releasing Phase 3 Data (currently, Novavax, Sanofi, and Bharat/Ocugen are all close to P3 results and EUA application) ✅ DONE 😉
3. FDA Approval
1. Going from the binding Letter of Intent (LOI) to the Definitive Agreement (DA) which should materialize at anytime. ✅ DONE 😉
2. Completing and releasing Phase 3 Data (currently, Novavax, Sanofi, and Bharat/Ocugen are all close to P3 results and EUA application) ✅ DONE 😉
3. FDA Approval
Step 1: ✅ DONE 😉 (now for Step 2 ⬇️)
Going from the binding Letter of Intent (LOI) to the Definitive Agreement (DA) which should materialize at anytime as the announcement was made on December 22, 2020 and the 3rd paragraph mentions,
Going from the binding Letter of Intent (LOI) to the Definitive Agreement (DA) which should materialize at anytime as the announcement was made on December 22, 2020 and the 3rd paragraph mentions,
- The companies have begun collaborating and will finalize details of the definitive agreement in the next few weeks.
- Announced that the companies have signed a binding letter of intent (LOI) to co-develop Bharat Biotech’s COVID-19 vaccine candidate, COVAXIN"
- This collaboration leverages Ocugen’s vaccine expertise, and its R&D and regulatory capabilities in the US.
- Ocugen will have US rights to the vaccine candidate and, in collaboration with Bharat Biotech, will be responsible for clinical development, registration, and commercialization for the US market
Step 2: ✅ DONE 😉 (now for Step 3 ⬇️)
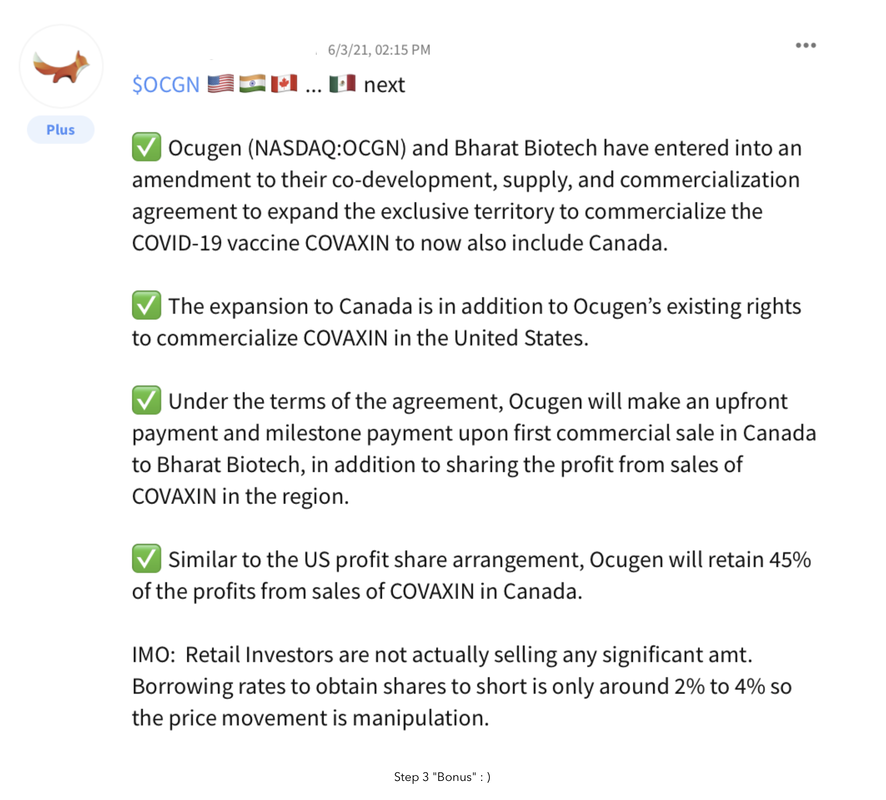
Completing and releasing Phase 3 Data (currently, Novavax, Sanofi, and Bharat/Ocugen are all close to P3 results and EUA application)
Completing and releasing Phase 3 Data (currently, Novavax, Sanofi, and Bharat/Ocugen are all close to P3 results and EUA application)
- COVAXIN has been evaluated in approximately 1,000 subjects in Phase 1 and Phase 2 clinical trials in India, with promising safety and immunogenicity data. The vaccine candidate is currently part of a Phase 3 clinical trial in India involving 26,000 volunteers
- In preparation for the development of COVAXIN in the US, Ocugen has assembled a Vaccine Scientific Advisory Board featuring leading academic and industry experts to evaluate the clinical and regulatory path to approval in the US market.
Step 3:
FDA Approval
FDA Approval
- “COVAXIN utilizes a historically proven approach to vaccine design. The adjuvanted inactivated virus vaccine candidate elicited strong IgG responses against spike (S1) protein, receptor-binding domain (RBD) and the nucleocapsid (N) protein of SARS-CoV-2 along with strong cellular responses in Phase 1 and 2 clinical trials. COVAXINTM offers a vaccine candidate that is different from other options currently available in the US market with potentially broader coverage against multiple protein antigens of the virus,” said Harvey Rubin, M.D. Ph.D. of the University of Pennsylvania, a member of Ocugen’s Vaccine Scientific Advisory Board.
- David Fajgenbaum, M.D. of University of Pennsylvania’s Division of Translational Medicine & Human Genetics, Director of the Center for Cytokine Storm Treatment & Laboratory, and member of Ocugen’s Vaccine Scientific Advisory Board said, “The COVID-19 pandemic has caused unmatched devastation to individual patients and to the world. It is going to take the kind of unmatched collaboration and innovation that is occurring right now to effectively fight back. Vaccines such as COVAXIN that can potentially elicit a broad immune response and may limit future COVID-19 severity could be important to have in our arsenal.”
- “We are delighted to collaborate with Bharat Biotech to potentially bring COVAXINTM to the US market. In the face of the coronavirus pandemic, it is incumbent upon all of us to find solutions that have the potential to save lives and restore normalcy to our day-to-day activities. We have been very pleased with the safety and immunogenicity demonstrated by the Phase 1 and Phase 2 trials of COVAXIN and are encouraged with the progress of the Phase 3 trials in India. We believe this unique yet traditional approach to vaccination holds great potential to appeal to a broad range of the population,” said Dr. Shankar Musunuri, Chairman, CEO, and Co-Founder of Ocugen.
- “The development and clinical evaluation of COVAXIN marks a significant milestone for vaccinology in India. COVAXIN has garnered interest from several countries worldwide for supplies and introduction and we are excited to collaborate with Ocugen to bring it to the US market,” said Dr. Krishna Ella, Chairman & Managing Director of Bharat Biotech.
Important Hyperlinked Post
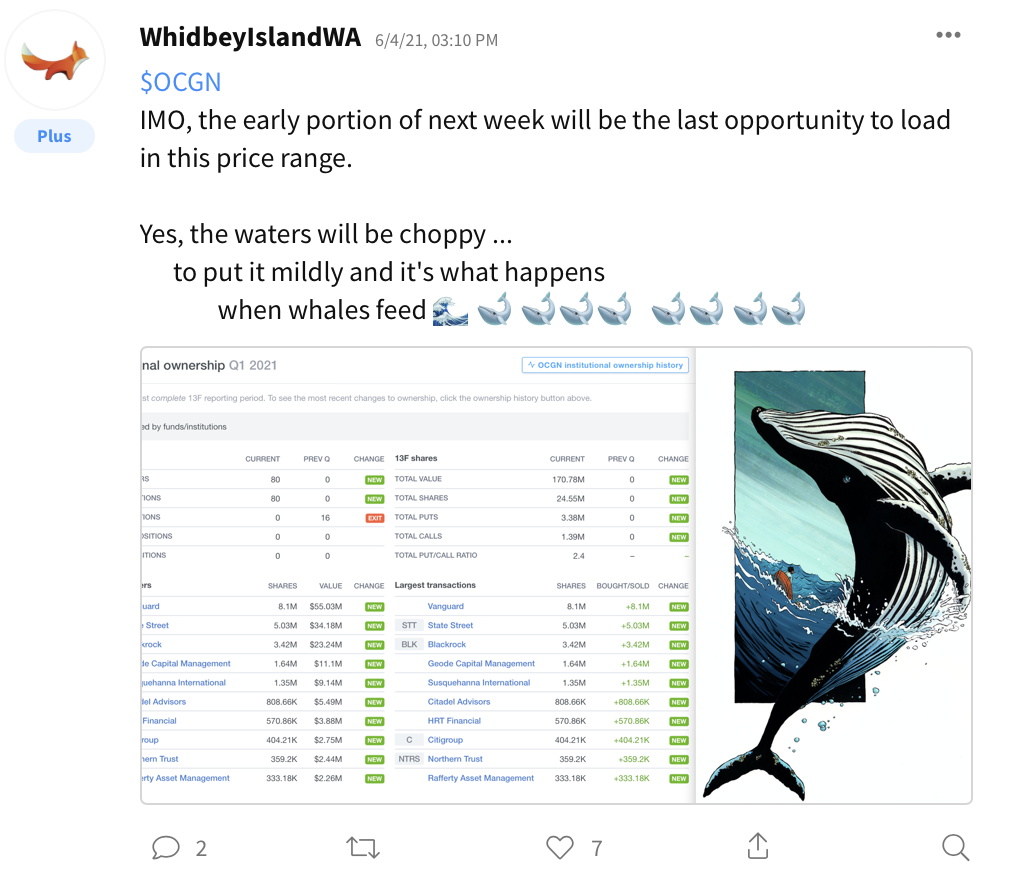
Stocktwits Users to Follow for OCGN
News Items
Misc.
Quick Links:
Hello 🙋🏻♀️, fellow OCGN Investors!
This webpage has been put together for you!
Please help by providing me w/ feedback, important news items others will benefit from, as well as, users we should follow specific for OCGN. I will add such items to this webpage.
You are welcome to direct others here and copy/paste and post so that we can collaboratively reduce repeated questions or misunderstandings by providing answers w/ links. Additionally, this webpage will help reduce FUD (fear, uncertainty, and doubt), as ppl will attempt to short OCGN.
I appreciate your assistance! 😀
This webpage has been put together for you!
Please help by providing me w/ feedback, important news items others will benefit from, as well as, users we should follow specific for OCGN. I will add such items to this webpage.
You are welcome to direct others here and copy/paste and post so that we can collaboratively reduce repeated questions or misunderstandings by providing answers w/ links. Additionally, this webpage will help reduce FUD (fear, uncertainty, and doubt), as ppl will attempt to short OCGN.
I appreciate your assistance! 😀