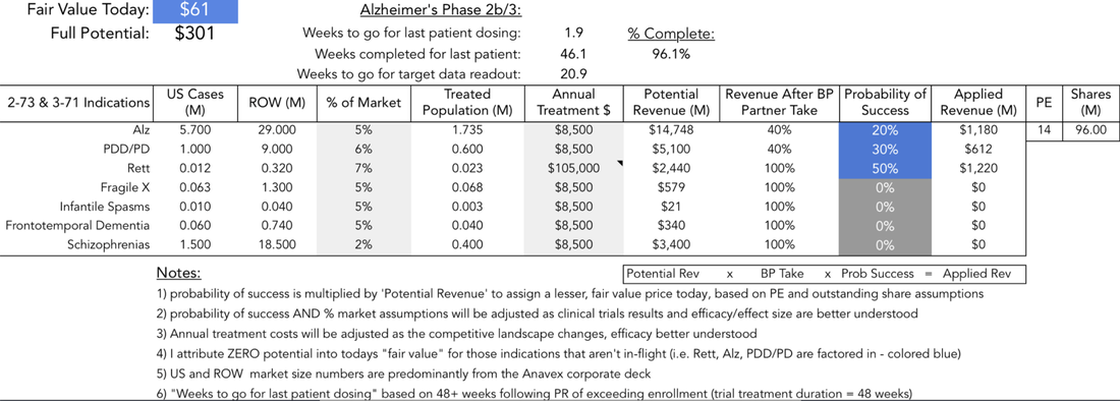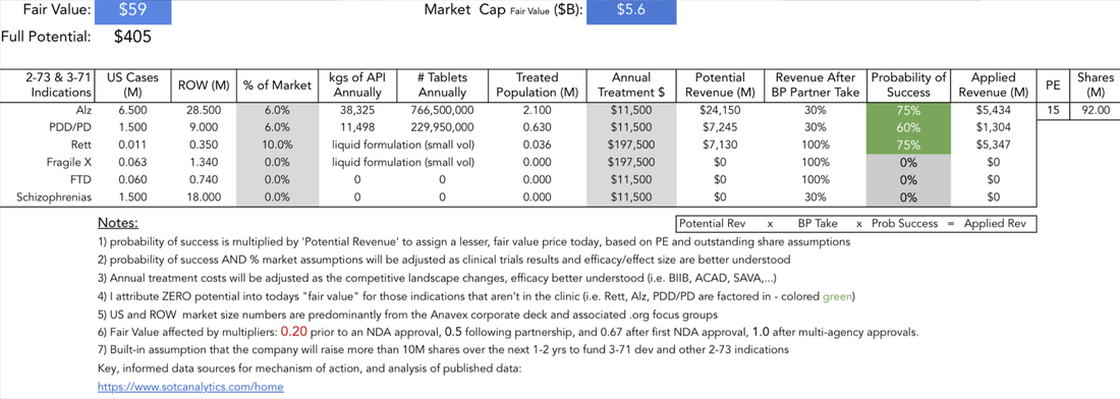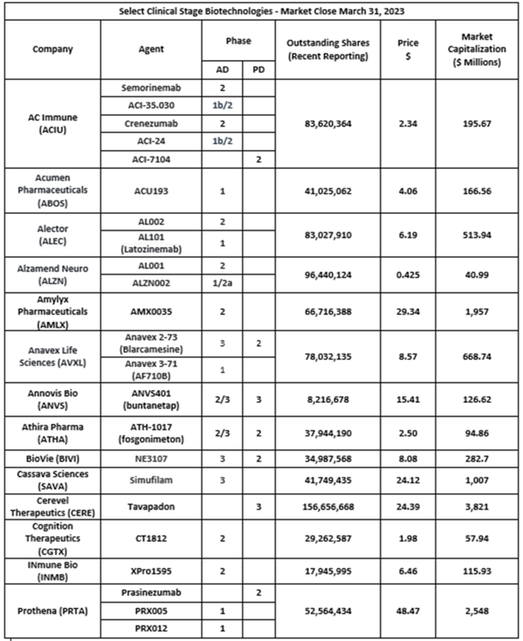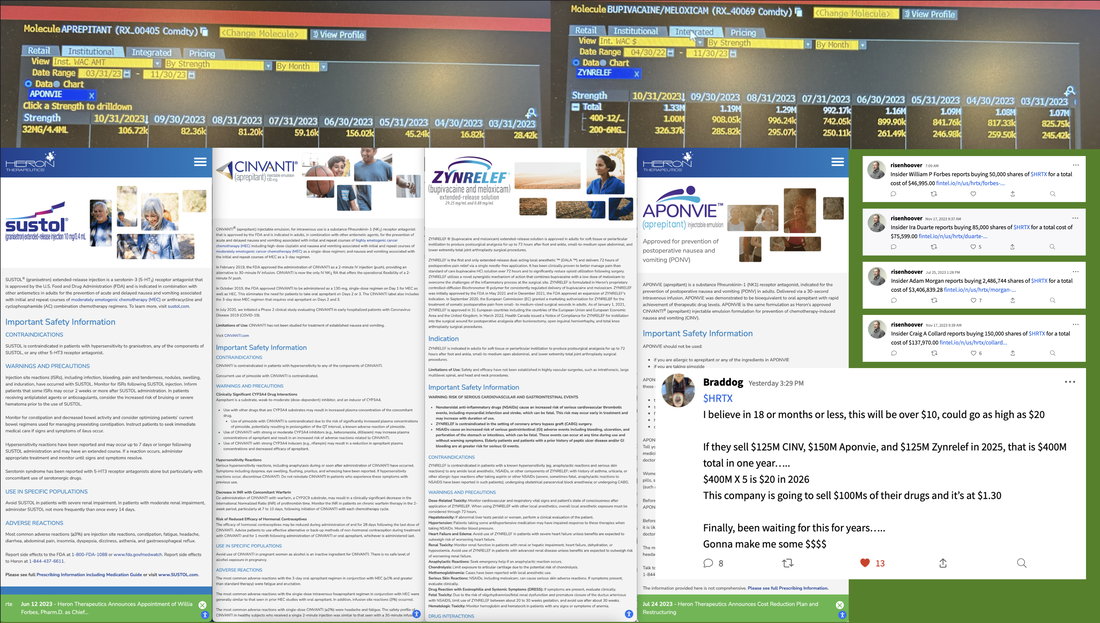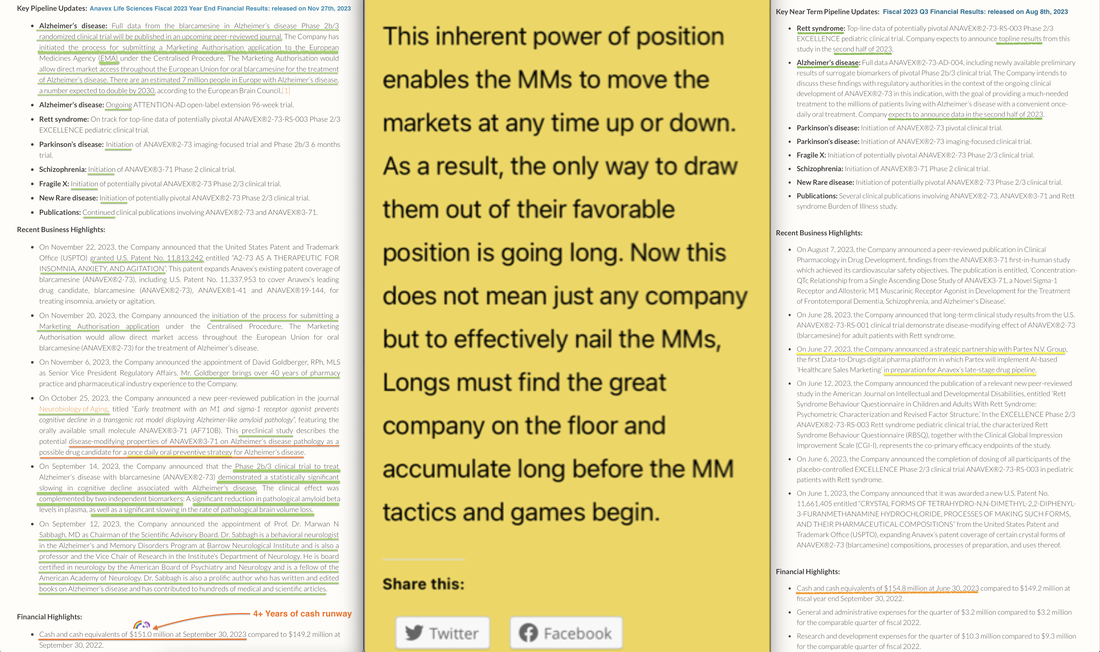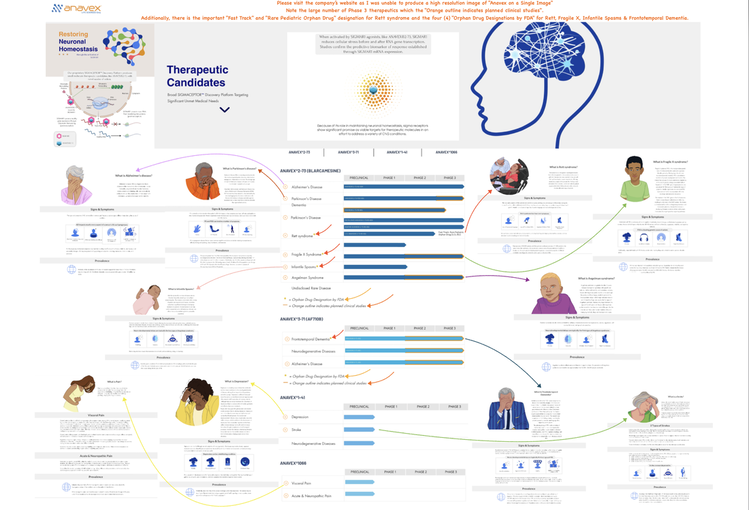
I was not able to create a single readable image given all the therapeutic candidates that Anavex is currently studying. For information on each of these diseases, please go the Anavex's website and click "Our Dedication". Thank you. (Alternately, click the image above to see a similar image in PDF format.)
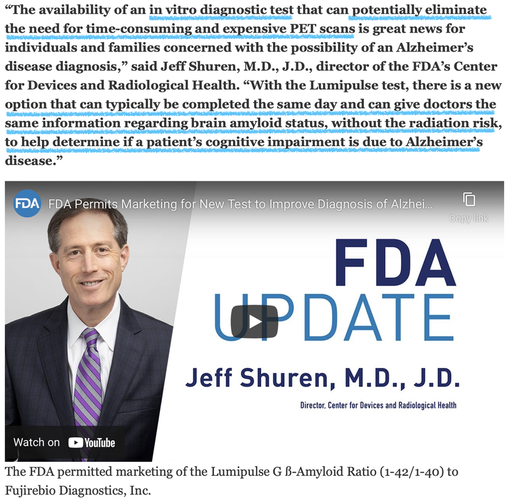
The U.S. Food and Drug Administration today permitted marketing for the first in vitro diagnostic test for early detection of amyloid plaques associated with Alzheimer’s disease. The Lumipulse G β-Amyloid Ratio (1-42/1-40) test is intended to be used in adult patients, aged 55 years and older, presenting with cognitive impairment who are being evaluated for Alzheimer’s disease and other causes of cognitive decline.