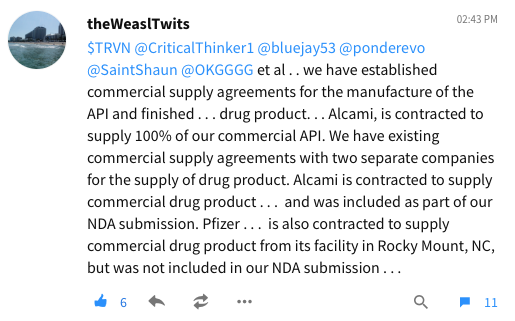
Alcami Master Commercial Supple Agreement 🇺🇸
https://www.trevena.com/investors/financial-information/annual-reports/content/0001104659-18-040166/a18-15338_1ex10d45.htm?TB_iframe=true&height=auto&width=auto&preload=fals
https://www.trevena.com/investors/financial-information/annual-reports/content/0001104659-18-040166/a18-15338_1ex10d45.htm?TB_iframe=true&height=auto&width=auto&preload=fals
|
(NOT PRESENTLY UTILIZED)
Source & Date: SEC, date of report 2019.12.04 (Pfizer production agreement : NC Facility) Title: Form 8-K Trevena Inc, current report, items 1.01 and 9.01 https://sec.report/Document/0001104659-19-070807/ Amendment No. 2 to Development and Supply Agreement, by and between the Company and Pfizer CenterOne Group of Pfizer Inc. dated as of December 2, 2019 https://sec.report/Document/0001104659-19-070807/ |
|
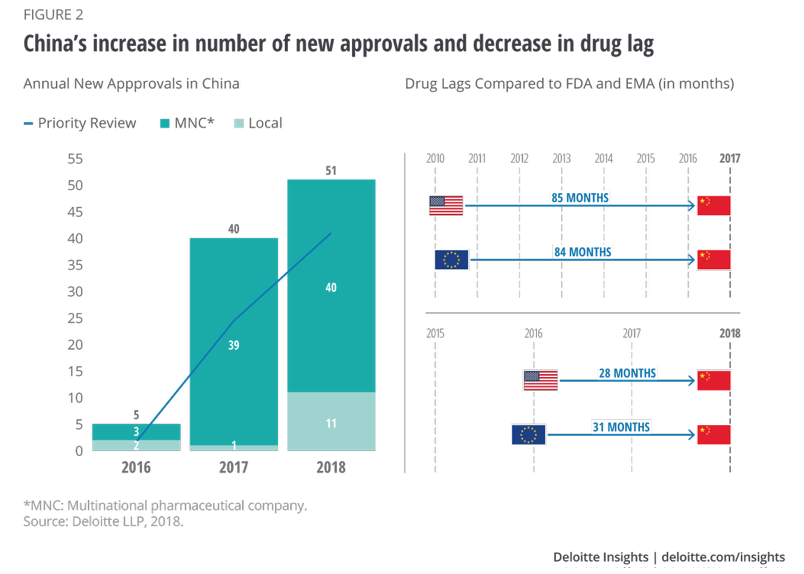
According to this study by Deloitte, the lag between US approval and an approval by China has been reduced to 28 months as of 2018. There appears to be motivation shorten this even more, "The reforms brought a fast-track approval process and a potential local-study waiver for products targeting rare diseases or diseases with substantial unmet needs.6 Since then, China has witnessed exponential growth in new approvals, and a significant reduction in drug lag as compared to the US Food and Drug Administration (FDA) and European Medicines Agency (EMA), as shown in Figure 2."
Source & Date: Science Direct 2015.03 "Today, China represents the 2nd largest health care market globally. China is the No. 2 market right now with lots of patients and in a few years, it will be the No. 1 market" from Apr 2020 article
Title: Heart Failure Therapeutics on the Basis of a Biased Ligand of the Angiotensin-2 Type 1 Receptor: Rationale and Design of the BLAST-AHF Study (Biased Ligand of the Angiotensin Receptor Study in Acute Heart Failure) |
Source & Date: Globe News wire 2018.05.01
Title: Trevena Inc. and Jiangsu Nhwa Pharmaceutical Co. Ltd Announce License Agreement for Oliceridine in China 🇨🇳
dun & bradstreet link: Jiangsu Nhwa Pharmaceutical Co., Ltd is located in Jiangsu, China and is part of the Pharmaceutical Manufacturing Industry. Jiangsu Nhwa Pharmaceutical Co., Ltd has 4,168 employees across all of its locations. There are 10 companies in the Jiangsu Nhwa Pharmaceutical Co., Ltd corporate family.
Title: Trevena Inc. and Jiangsu Nhwa Pharmaceutical Co. Ltd Announce License Agreement for Oliceridine in China 🇨🇳
dun & bradstreet link: Jiangsu Nhwa Pharmaceutical Co., Ltd is located in Jiangsu, China and is part of the Pharmaceutical Manufacturing Industry. Jiangsu Nhwa Pharmaceutical Co., Ltd has 4,168 employees across all of its locations. There are 10 companies in the Jiangsu Nhwa Pharmaceutical Co., Ltd corporate family.
Source & Date: Globe News wire 2018.04.27
Title: Trevena and Pharmbio Korea Announce License and Commercialization Agreement for Oliceridine in South Korea 🇰🇷
Title: Trevena and Pharmbio Korea Announce License and Commercialization Agreement for Oliceridine in South Korea 🇰🇷
|
FDA "Changes to an Approved NDA or ANDA"
https://www.fda.gov/media/71846/download DEA "Change Drug Scheduling" is possible (see 2nd paragraph) https://www.dea.gov/controlled-substances-act Trevena Job Openings: https://www.trevena.com/contact/careers Syneos Health Openings: https://commercialcareers.syneoshealth.com/jobs/search?location=&page=1&q=Trevena# |